News Release
UC San Diego team develops new point-of-care analyzer for human milk
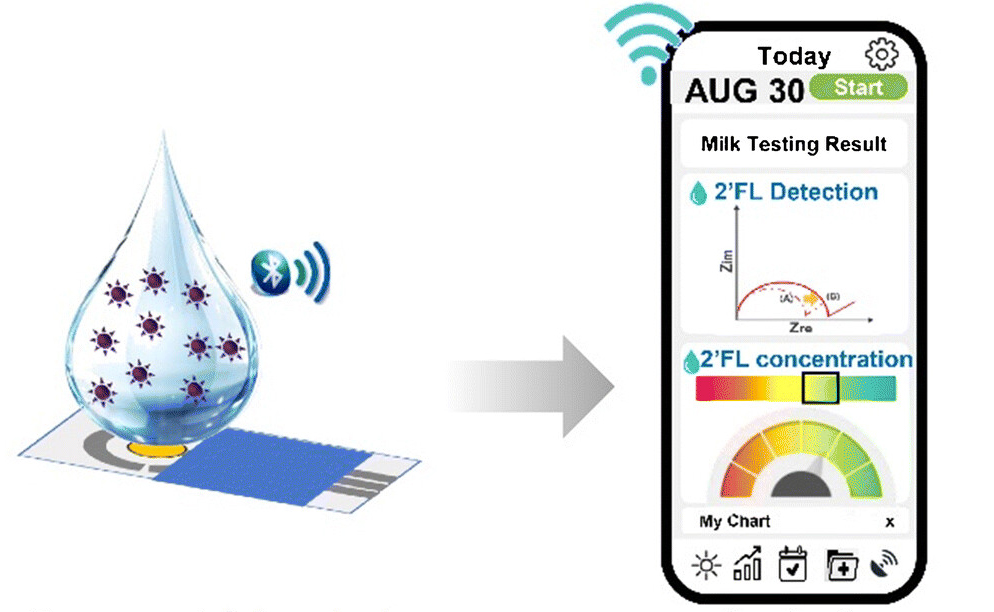 |
| Illustration of proposed human milk assay. Image adapted from Analytical and Bioanalytical Chemistry |
November 10, 2021 -- Measuring human milk oligosaccharides (HMOs) at the point of care, in the NICU, in milk banks, at home or even on your cell phone may soon be possible, thanks to a new device developed by researchers at the University of California San Diego.
The device is a biosensor that tests for maternal secretor status and measures concentrations of the HMO 2’-fucosyllactose (2’FL) in a drop of human milk in just 35 minutes.
The work, led by postdoctoral researcher Saeromi Chung and Professor Drew Hall in the Department of Electrical and Computer Engineering at UC San Diego, is described in a paper published Nov. 5 in the journal Analytical and Bioanalytical Chemistry. The team included Lars Bode, a professor of pediatrics at UC San Diego School of Medicine and director of the Larsson-Rosenquist Foundation Mother-Milk-Infant Center of Research Excellence (MOMI CORE).
High concentration of 2’FL in human milk has been linked to infant growth, improved neurodevelopment, and protection from infectious diarrhea, which is one of the major causes of infant death worldwide. However, there is currently no technology to analyze the presence and concentration of HMOs such as 2’FL in human milk at the point of care.
The lack of such technology “represents a major impediment to advancing human milk research and improving maternal-infant health,” the researchers wrote.
The new technology developed at UC San Diego will be a game changer for research, donor milk banking and clinical applications, the researchers said. It consists of a simple biosensor, made of an electrode containing molecules that selectively bind to 2’FL. Place a drop of human milk sample on the electrode tip and within 35 minutes, it produces an electrochemical readout of the 2’FL concentration.
The team is improving the device so that it can communicate with and display readings on smartphones.
Paper: “Point-of-care human milk testing for maternal secretor status.”
This work was supported by pilot grants from the MOMI CORE Seeds program and the Altman Clinical and Translational Research Institute (ACTRI) at UC San Diego.
Media Contacts
Liezel Labios
Jacobs School of Engineering
858-246-1124
llabios@ucsd.edu